 It’s useful in some special medical conditions that require the decent support of anesthesia.History Discovered In 1898 Discovered By Sir William Ramsay, Morris W. 
Xenon anesthesia is generally much more expensive than regular anesthesia. 54 electrons (white) occupy shells (rings) eight fill the outer (fifth) electron shell in what is a very stable configuration. It’s primarily used in the production of unique kinds of anesthesia. The first is to use the Periodic Table to figure out how many electrons Xenon has i. The Xenon gas light is highly powerful and is useful even in space ships or rockets in space travel.įurthermore, Xenon is also useful in the medical or pharmaceutical industry. There are two ways to find the number of valence electrons in Xenon (Xe). Explanation: The element is defined by Math Processing Error, the atomic number, which is the number of protons, positively charged, massive nuclear particles. The primary usage of Xenon lies in the production of light-emitting devices such as flashlights, vehicle headlights, etc. 1 Answer anor277 The electronic configuration is there to distract you: Math Processing Error, and, therefore, the element is LEAD. 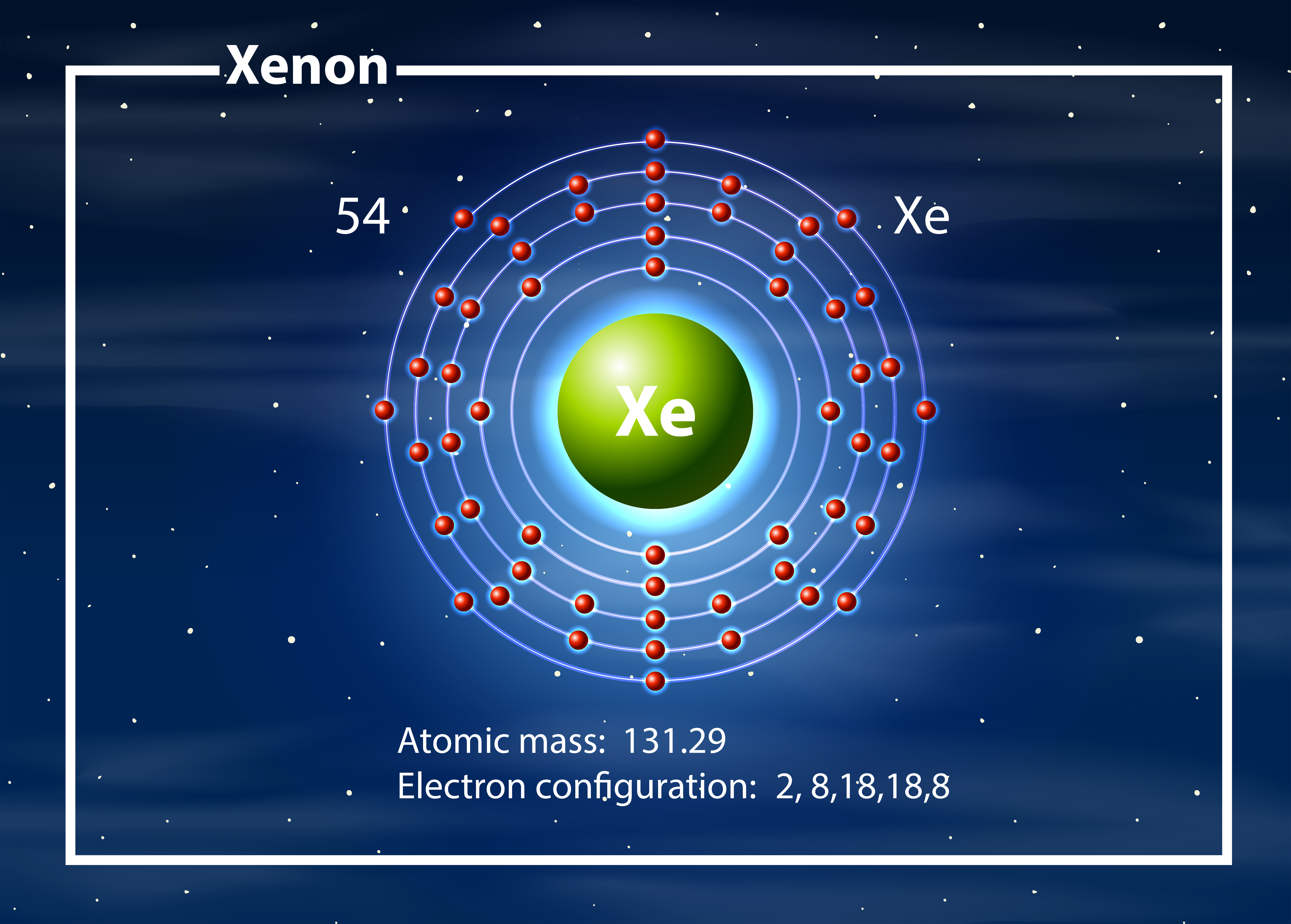 
Well, Xenon is a highly precious noble gas in the domain of the lighting industry around the world and remains in significantly high demand. It can be found in group 18, and it has an atomic symbol, Xe, and an atomic mass of 131.293 u. There are several other properties of Xenon that you can explore in the periodic table of the element. Xenon has an electronic configuration of Kr 4d10 5s2 5p6. It subsequently helps in finding the other legitimate usages of the Xenon in chemistry. For instance, with the help of electron configuration, one can easily get to understand the respective reaction of elements with the other elements. The I ion is a iodine atom that has acquired one extra electron therefore, it has an electron configuration the same as xenon. The electron configuration of the Xenon reveals the other major characteristics or properties of the element. The abbreviated electronic configuration of Xenon is Kr 4d10 5s2 5p6. The electron configuration of Xenon with the same principle is 4d¹⁰ 5s² 5p⁶ for the reference of our scholars. How many valence electrons does Xenon have? We know that xenon's full electron configuration is 1s2\2s2\2p6\3s2\3p6\3d10\4s2\4p6\4d10\5s2\5p6 From this picture, we see that the next shell we choose to fill will be the 6s orbital. (a) Give the electron configurations (using the noble gas notation) for Ce, Ce3 , and Ce4 . Cerium (III) sulfide, Ce2S3, is becoming more widely used as a red pigment to replace cadmium pigments, which are environmentally less desirable. This distribution in results takes the shape of an equation that becomes the electron configuration of the Xenon. So, we start by writing the first part of tungsten's electron configuration which is Xe Next, we have 74-5420 more electrons to fill. CeO2, is widely used as a polishing agent for glass. For the same purpose, we basically distribute the electrons of Xenon to its orbitals for the distribution of electrons. The understanding of Xenon electron configuration is essential in order to understand this noble gas element in a thorough manner. 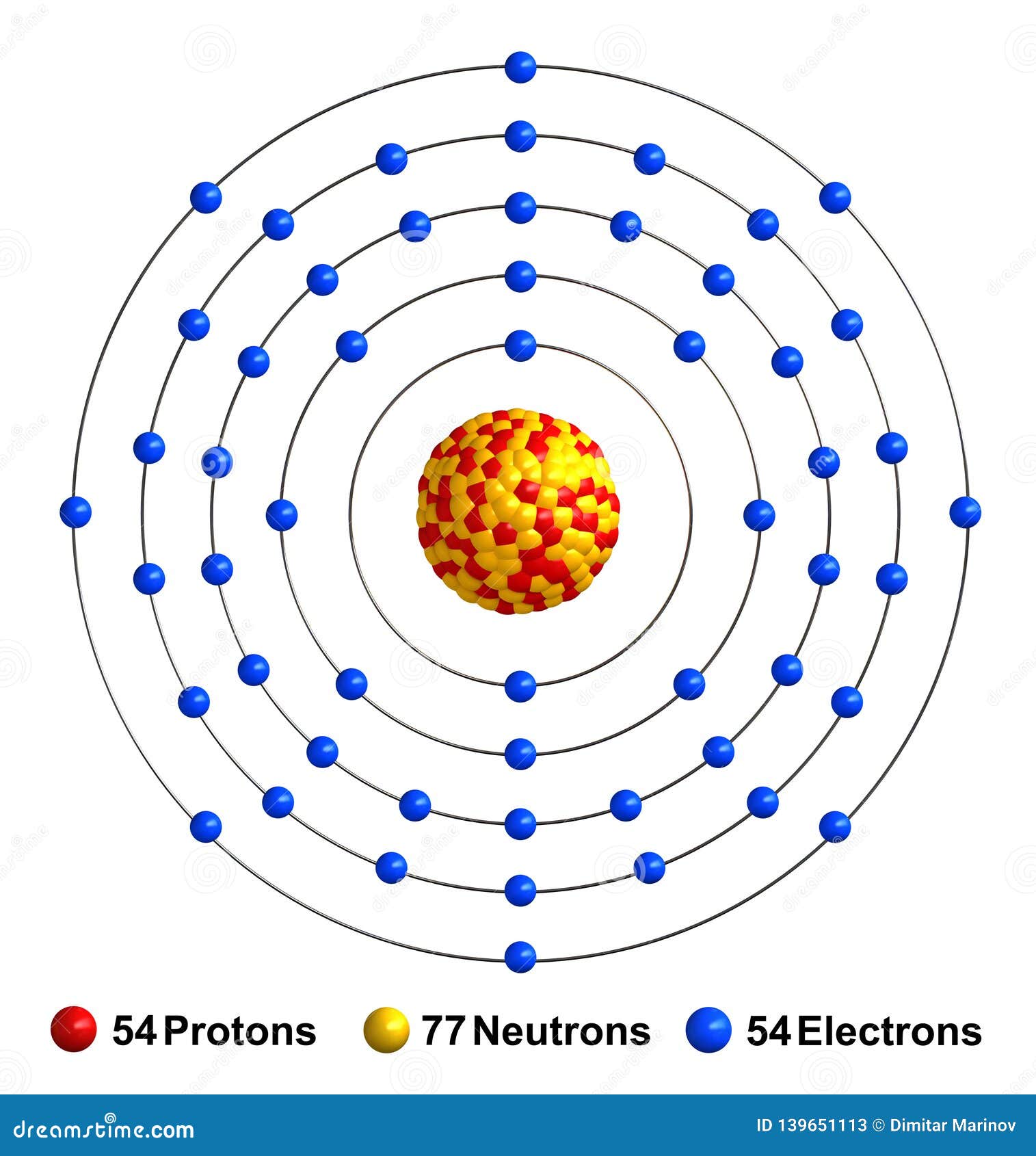
This only applies to the first row transition metals, adjustments will be necessary when writing the electron configuration for the other rows of transition metals. You can easily find this chemical element in the atmosphere of earth with a significant amount. The electron configuration for the first row transition metals consists of 4s and 3d subshells with an argon (noble gas) core. Xenon is precisely the gas that has the texture as the colorless, dense, and odorless liquid. It has the atomic number 54 and the representative sign as Xe. Xenon is a well-known chemical element in the chemistry domain of science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
